Atopic dermatitis doesn’t just affect your skin — it disrupts your whole day. And for half of adults with moderate to severe atopic dermatitis, it’s a major lifestyle burden.1
We’re conducting a clinical research study designed to test the safety and efficiency of a potential treatment option in reducing signs and symptoms of moderate to severe Atopic Dermatitis, also known as eczema.



*For additional eligibility criteria of the clinical trial, please visit: clinicaltrials.gov

The purpose of this clinical study is to determine how effective an investigational injectable medication is at treating moderate to severe atopic dermatitis, commonly known as eczema.
The investigational treatment has the potential to diminish the immune response associated with atopic dermatitis. The goal is to reduce inflammation, which may help ease symptoms of atopic dermatitis such as itching, dryness, cracked skin, and rashes.

Participants will be randomly assigned to one of two treatment groups. Some will receive the investigational injectable study drug, while others will get a matching placebo. A placebo contains no active treatment and is used to ensure that the changes you report are not due to chance. Often called a “dummy” injection, an injectable placebo usually contains a harmless substance like saline solution (salt water).
You do not have to pay for the investigational study drug or study procedures that are not part of your regular medical care. However, you may be responsible for paying for routine medical care that may be performed during the trial, some of which may be covered by your insurance.
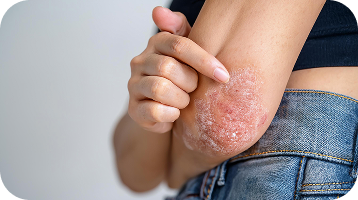
Atopic Dermatitis frequently appears in areas where your skin bends or flexes, such as behind the knees or inside the elbows; however, it can develop anywhere2. Signs and symptoms of this condition include3,4:

Persistent, intense itching (pruritus) that often worsens at night and drives scratching, triggering flare-ups and worsening overall inflammation.

Skin becomes rough, flaky, or tight, frequently cracking or peeling—especially over time or when scratched repeatedly.

Red, inflamed, swollen or scaly patches that vary by skin tone (reddish, brownish gray, or purple) and can feel hot, sore, or tight.

Tiny blisters (vesicles) may develop and ooze clear fluid; after bursting, they often crust or bleed and can lead to skin infections.
If you’re eligible and choose to enroll, you’ll be asked to participate in several visits throughout the study. These visits will involve traveling to a nearby study location, usually at healthcare facilities like hospitals or clinics, where you’ll participate in study-related activities.
During your visits, you might receive either the study drug or a placebo. You’ll need to undergo physical exams, have your vital signs checked, and have some laboratory tests to monitor and ensure your safety. All the details of study-related care will be clearly explained to you before you enroll in the study.




Answer a few short questions online to determine if you meet the initial eligibility criteria to join a study near you.
If you meet the initial criteria and grant permission, a Study Team representative will call you within two business days to ask additional screening questions and confirm the answers you have submitted online.
If you meet all the eligibility requirements, you’ll have the opportunity to visit a study site near you. There, you’ll undergo health checks, tests, and receive a full explanation of the study. If you decide to participate, you’ll review and sign the informed consent form.
If you qualify based on your screening results, you’ll be officially enrolled and randomly assigned either the study drug or a placebo. Neither you nor the study doctor will know which group you belong to. A placebo contains no active ingredients and is used to ensure that the changes you report are not due to chance.
1. National Eczema Association. “Eczema Stats,” February 27, 2025. https://nationaleczema.org/eczema-facts/.
2. Cleveland Clinic. “Atopic Dermatitis: Symptoms, Causes & Treatment,” October 18, 2022. https://my.clevelandclinic.org/health/diseases/24299-atopic-dermatitis.
3. King, Lori M. “What Is Atopic Dermatitis (Eczema)?” WebMD, June 26, 2016. https://www.webmd.com/skin-problems-and-treatments/eczema/eczema-basics.
4. “What Is Atopic Dermatitis?” Accessed July 11, 2025. https://nationaleczema.org/wp-content/uploads/2024/09/NEA_Factsheets_AD101Adults_D01.pdf
We do not sell data to third parties. Data you submit is handled according to our Privacy Policy and web site use agreements.
To control data you have already submitted, or to log a do not sell data request, make a data request here.
To control data you have already submitted, make a data request here.
Data you submit is handled according to our Privacy Policy and web site user agreements.